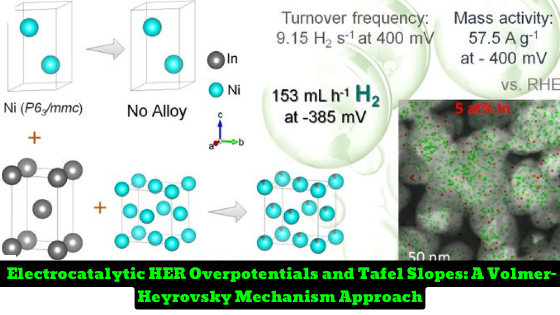
IISER Kolkata Researchers have developed a series of Ni-In heterogeneous alloys by addressing the lattice immiscibility between the two elements. The flower-like self-assembly of Ni NPs consists of 36 wt% fcc and 64 wt% hcp phases before the addition of indium. The addition of indium selectively increased the fraction of fcc Ni, leading to an increase in unit cell parameters and volume of the fcc phase while the hcp phase remains unaltered. The addition of indium led to a charge transfer from In to Ni and stabilized the metallic state of the Ni-sites. The best HER activity was observed with In5 with a TOF of 9.15 s-1 at -400 mV versus RHE and a mass activity of 57.5 A g-1 at -400 mV overpotential. The research has been published in the journal Angewandte International Edition Chemie.
The researchers investigated the electrocatalytic HER performance of the Ni-In heterogeneous alloys, In5, In10, and In15, and found that the adsorption energies of hydrogen and hydroxyl ions on In5 were optimum, leading to an increase in alkaline HER activity more than other systems. The HER overpotentials of In5 and In10 were 77 mV and 185 mV, respectively, at 10 mA cm-2. The electrochemical active surface area (ECSA) value was directly proportional to the double-layer capacitance (Cdl) in the non-faradaic region by varying the scan rate. The best-performing In5 catalyst was tested for chronoamperometric stability for 200 h at -0.18 V versus RHE.
The lattice mismatch-guided nickel-indium heterogeneous alloy electrocatalysts showed promising results for promoting alkaline hydrogen evolution. The addition of indium selectively increased the fcc phase fraction, leading to an increase in HER activity. The study provides insights into the arrangement of fcc and hcp phases in Ni-In alloys and their effects on HER activity. The findings of the study could be useful in designing efficient and stable electrocatalysts for hydrogen evolution.
Title: Lattice Mismatch Guided Nickel-Indium Heterogeneous Alloy Electrocatalysts for Promoting the Alkaline Hydrogen Evolution Lattice Mismatch Guided Nickel-Indium Heterogeneous Alloy Electrocatalysts for Promoting the Alkaline Hydrogen Evolution
Mondal; Dutta; Mal; Pati; Bhattacharyya; Mondal; Dutta; Mal; Pati; Bhattacharyya
Full-text link: https://doi.org/10.1002/anie.202301269
Help us improve! Select the sentence(s) if helpful, and click on “send feedback” below.
What this paper is about
- One way to stabilize the metallic state of nickel for catalyzing HER, is to design the alloy with a more oxophilic metal counterpart such as Ti.
- As a result, with increasing In at%, the hcp phase wt% of Ni is reduced at the cost of fcc Ni, and the intricacies of the arrangement of the fcc and hcp phases were elucidated by electron microscopy.
- In, shows the most prolific enhancement of the HER activity at pH 14, with a mass activity of 57.5 A g -1 at -400 mV overpotential, and a turnover frequency of 9.15 s -1 at -400 mV versus reversible hydrogen electrode.
What you can learn
- The powder X-ray diffraction pattern of the In0 sample shows two phases of Ni, fcc phase with the reflections at 2 = 44.
- Because of the exclusive incorporation of larger-sized In into the fcc phase of Ni, the corresponding 2 reflections are downshifted while that for the hcp phase remains unaltered.
- The selective alloying of In with fcc Ni can be explained by the possibility of a better solid solution when the dissolution of facets from different lattices is less compromised.
- Also, when the electrode has an active area of 0.25 cm, 153 mL h -1 H2 is consistently produced at -385 mV versus RHE.
- In comparison to In5, only Ni needs = 350 mV at 10 mA cm -2.
- With higher In in the heterogeneous alloys, the HER activity is gradually suppressed as shown by = 77, 185 and 268 mV at 10 mA cm -2 for In10.
What is the focus of the research discussed in the given paragraphs?
The research focuses on the creation of a series of Ni-In heterogeneous alloys through a non-aqueous heat-up colloidal reduction process, and investigating their electrocatalytic hydrogen evolution reaction (HER) performance. The research also addresses the lattice immiscibility between Ni and In, and how it affects the HER activity of the resulting alloys. The study explores the role of In incorporation in increasing the fcc phase of Ni, and how it stabilizes the metallic state of Ni for catalyzing HER. The research also delves into the adsorption of hydrogen and hydroxyl ions on In5, and how it helps in increasing the alkaline HER activity.
What is lattice immiscibility?
Lattice immiscibility refers to the inability of two metals to form a homogeneous solid solution due to significant differences in their lattice structures or crystallographic parameters.
What are heterogeneous alloys?
Heterogeneous alloys refer to alloys that consist of two or more different types of metals or elements, which have different crystal structures, atomic sizes, or electronegativities.
What is the non-aqueous heat-up colloidal reduction process?
The non-aqueous heat-up colloidal reduction process is a chemical method used to synthesize nanoparticles in a non-aqueous solvent by reducing metal salts with a reducing agent under elevated temperatures.
What are fcc and hcp phases?
Fcc (face-centered cubic) and hcp (hexagonal close-packed) are two common crystal structures of metals. The fcc structure has atoms at the corners and center of each face of a cube, while the hcp structure has atoms arranged in a close-packed hexagonal lattice.
What is lattice mismatch?
Lattice mismatch refers to the difference in the lattice parameters or crystal structures between two materials, which can cause strain and defects at the interface when they are joined together.
What are unit cell parameters?
Unit cell parameters are the dimensions and angles that define the repeating unit of a crystal lattice, including the length of the edges, the angles between them, and the position of atoms within the cell.
What is charge transfer?
Charge transfer refers to the process of transferring electrons or ions from one species to another during a chemical reaction or an electrochemical process.
What is Bader charge analysis?
Bader charge analysis is a method used to calculate the electronic charge density and the atomic charges of individual atoms in a crystal structure based on the quantum mechanical calculations.
What is alkaline HER activity?
Alkaline HER activity refers to the ability of a catalyst to promote the hydrogen evolution reaction (HER) in an alkaline environment, which is an important reaction in electrocatalysis and energy conversion.
What is electrocatalytic?
Electrocatalytic refers to the ability of a material or substance to catalyze or promote an electrochemical reaction, such as the conversion of chemical energy into electrical energy or vice versa.
What are HER overpotentials?
HER overpotentials refer to the additional potential or voltage needed to drive the hydrogen evolution reaction (HER) at a certain rate, above the equilibrium potential of the reaction.
What are Tafel slopes?
Tafel slopes refer to the slope of the linear relationship between the logarithm of the current density and the applied potential in an electrochemical reaction, which provides information about the reaction mechanism and the kinetics of the reaction.
What is Volmer-Heyrovsky mechanism?
Volmer-Heyrovsky mechanism is a two-step mechanism proposed for the hydrogen evolution reaction (HER), involving the adsorption and reduction of protons on the surface of a catalyst, followed by the desorption of hydrogen molecules.
What is electrochemical active surface area (ECSA)?
Electrochemical active surface area (ECSA) refers to the area of the surface of an electrode that is electrochemically active and participates in the electrochemical reaction, which is an important factor in determining the electrocatalytic activity of a catalyst.
What is chronoamperometric stability test?
Chronoamperometric stability test is an electrochemical method used to evaluate the stability and durability of a catalyst or electrode by measuring the current response over time under constant potential or current conditions.
What is Volmer-Heyrovsky mechanism?
Volmer-Heyrovsky mechanism is a two-step mechanism proposed for the hydrogen evolution reaction (HER