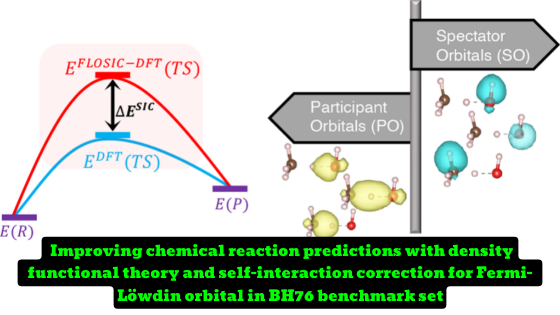
A new study conducted by researchers has delved into the origin of one-electron self-interaction errors (SIE) in density functional theory (DFT) predictions of chemical reaction barrier heights. The study analyzed data from the Fermi–Löwdin orbital self-interaction correction (FLOSIC) calculations to understand the SIE contributions to the reactions in the BH76 benchmark set. The study found that the largest source of one-electron SIE came from the most delocalized participant orbitals (POs) in the transition state (TS) of a reaction.
The study also hypothesized that the stretched bond orbitals would account for most of the correction for a given barrier. However, this hypothesis did not hold for either PZSIC or LSIC for the BH76 reactions. For reactions where the total SIC contribution to the barrier was larger than 10 kcal/mol, the hypothesis held in 79% of reactions in PZSIC but only 45% of reactions in LSIC. The overall agreement of the PZSIC and LSIC reaction barriers with reference values was better when contributions from both the POs and spectator orbitals (SOs) were included.
The results of the study also showed that the contribution of the density functional approximation (DFA) part of the total energy to the barrier height error was 5.2, and this contributed to the better overall performance of LSIC for the BH76 data set.
Self-interaction corrections can be viewed as addressing either functional-driven or density-driven errors in a DFA. The study found that the largest SIC contribution to barrier height corrections stems from the delocalized stretched bond orbitals, and there is an energy penalty for the stretched bond POs in the transition state in PZSIC–LSDA calculations. The study has provided new insights into the origin of one-electron self-interaction errors in DFT predictions of chemical reactions and suggests that further studies in this field are necessary to improve the accuracy of DFT predictions.
How Do Self-Interaction Errors Associated with Stretched Bonds Affect Barrier Height Predictions?
Shukla; Mishra; Baruah; Zope; Jackson; Johnson 2023
Full-text link: https://doi.org/10.1021/acs.jpca.2c07894
What this paper is about
- Because PZSIC and LSIC are orbital-by-orbital correction schemes, the contributions of individual orbitals to the overall correction of chemical barrier heights can be determined.
- The analysis consists of identifying the POs and SOs in the transition state and the corresponding orbitals in the reactant or product states and comparing the SIC energies of these orbitals to determine their contributions to the overall reaction barrier correction.
- Our analysis also shows that LSIC predicts barriers more accurately than PZSIC where the overall barrier correction is small and that this accounts for the overall better performance of LSIC for the BH76 set.
What you can learn
- For PZSIC, we find that the hypothesis is satisfied in 79% of the reactions where E Total SIC > 10 kcal/mol, but in only 52% where E Total SIC < 10 kcal/mol.
- For LSIC), the hypothesis is followed in 45% of reactions where the barrier correction is large and in 58% when the correction is small.
- The PZSIC contribution to the reaction barrier for the for T4 reaction is E Total SIC = 17.19 kcal/mol, placing T4 in the group of reactions with large barrier corrections.
- By comparing the SIC energy for an orbital in the reactants/products and for the corresponding orbital in the transition state of a given reaction, we obtain the SIC contribution to the forward/reverse reaction barrier coming from that orbital.
- The SIC energy for this stretched bond orbital is considerably more positive than for the corresponding orbital in the R or P. The correction to a barrier from this orbital is therefore positive, i.e., increasing the LSDA prediction for the barrier, which is nearly always too small.
- For reactions where the total SIC contribution to the barrier is larger than 10 kcal/mol, the hypothesis is valid in 79% of reactions in PZSIC but in only 45% of reactions in LSIC.
What is FLOSIC and how is it used in this work?
FLOSIC stands for Fermi-Löwdin orbital self-interaction correction and is a computational method used in this work to obtain orbitals that minimize the Perdew-Zunger total energy. The FLOSIC scheme is used to correct self-interaction errors in density functional theory (DFT) predictions of chemical reaction barrier heights. The PZSIC-LSDA and LSIC-LSDA functionals used in this work are exact for any one-electron density and can thus be said to be one-electron self-interaction-free. By removing self-interaction errors, the mean absolute error in the barrier prediction for the BH76 reactions dropped from 15.5 kcal/mol in LSDA to 5.4 and 3.7 kcal/mol in PZSIC-LSDA and LSIC-LSDA, respectively.
Q: What are Fermi-Löwdin orbitals?
A: Fermi-Löwdin orbitals are a set of orbitals that are constructed by diagonalizing the one-particle density matrix.
Q: What is self-interaction correction?
A: Self-interaction correction is a theoretical correction applied to density functional theory calculations to account for the overestimation of electron self-interaction.
Q: What is density functional theory?
A: Density functional theory is a theoretical framework used in chemistry and physics to describe the electronic structure of many-body systems, such as atoms, molecules, and materials.
Q: What is a chemical reaction?
A: A chemical reaction is a process in which one or more substances are converted into one or more different substances.
Q: What are barrier heights?
A: Barrier heights are the energy barriers that need to be overcome to transform reactants into products in a chemical reaction.
Q: What is the BH76 benchmark set?
A: The BH76 benchmark set is a set of 76 chemical reactions that is commonly used to test the accuracy of density functional theory calculations.
Q: What is LSDA?
A: LSDA stands for Local Spin Density Approximation, which is a common exchange-correlation functional used in density functional theory calculations.
Q: What is PZSIC?
A: PZSIC stands for Perdew-Zunger Self-Interaction Correction, which is a theoretical correction applied to density functional theory calculations to account for electron self-interaction.
Q: What is LSIC?
A: LSIC stands for Löwdin-Szabo Self-Interaction Correction, which is a theoretical correction applied to density functional theory calculations to account for electron self-interaction.
Q: What is mean signed error?
A: Mean signed error is a statistical measure used to evaluate the accuracy of a calculation, which measures the average difference between the calculated and actual values, with positive and negative errors canceling each other out.
Q: What is mean absolute error?
A: Mean absolute error is a statistical measure used to evaluate the accuracy of a calculation, which measures the average absolute difference between the calculated and actual values.
Q: What is SIE?
A: SIE stands for Self-Interaction Error, which is the error that arises in density functional theory calculations due to the overestimation of electron self-interaction.
Q: What is a transition state?
A: A transition state is a hypothetical species that represents the highest energy point along the reaction pathway, where the reactants are partially converted into products.
Q: What is bond-breaking?
A: Bond-breaking is the process of breaking chemical bonds in a molecule.
Q: What is bond-making?
A: Bond-making is the process of forming new chemical bonds between atoms.
Q: What are participant orbitals?
A: Participant orbitals are orbitals that are